No products in the cart.
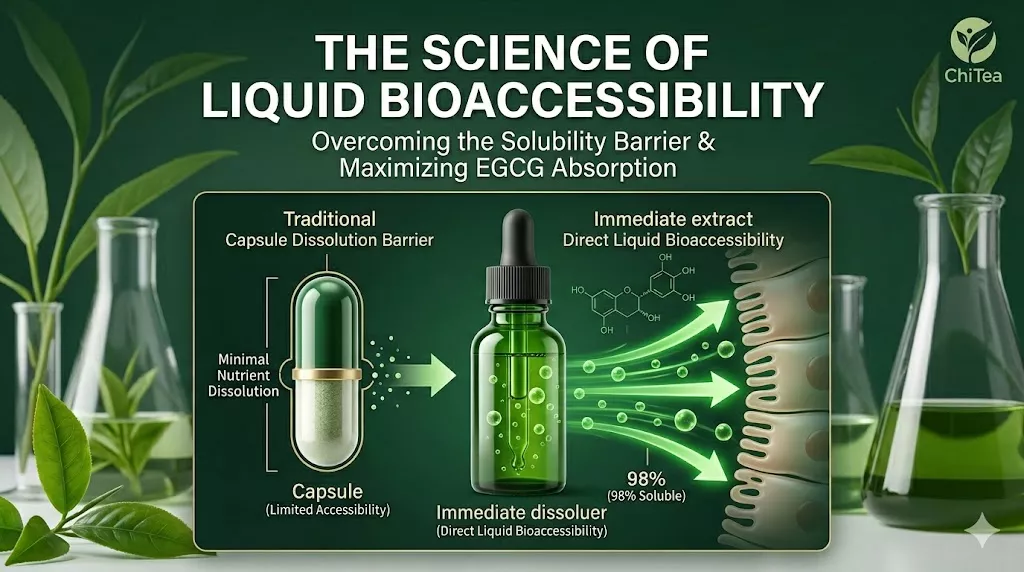
When evaluating the effectiveness of liquid green tea extract absorption, the most critical factor is not just the total dose, but the bioaccessibility of the molecules. Unlike traditional solid-dose formats that must undergo mechanical breakdown, ChiTea’s liquid protocol is pre-solubilized to bypass the “Solubility Barrier” common in encapsulated supplements. This ensures that the standardized 72mg of EGCG is immediately ready for intestinal transport. View peer-reviewed research on the factors affecting EGCG bioavailability.
In nutritional science, the effectiveness of a compound is limited by its Dissolution Rate. For a molecule like EGCG to be absorbed in the small intestine, it must first be fully dissolved in gastric fluid. Capsules and tablets face a ‘Solubility Barrier,’ where much of the active payload passes through the system before it can be solubilized. ChiTea’s liquid protocol removes this barrier by delivering 95% polyphenols in a pre-solubilized state, ensuring immediate bioaccessibility.
| Molecular Marker | Standardized Potency | Clinical Significance |
|---|---|---|
| EGCG (Epigallocatechin gallate) | 72mg per serving | The primary bioactive catechin for mitochondrial protection and cellular signaling. |
| Total Catechins | 80% (Standardized) | Full-spectrum green tea polyphenols for systemic antioxidant fortification. |
| Total Polyphenols | 95% (Standardized) | Maximum concentration of scavenger molecules to neutralize oxidative stress. |
| Solvent Residue | 0.00% (Pure) | Alcohol-free and solvent-free steam-water extraction process. |
| Heavy Metals / Microbial | Verified Negative | Independently tested for purity, ensuring safety for long-term clinical protocols. |
| Efficiency Metric | ChiTea Liquid Protocol | Solid Dose (Capsules/Tablets) |
|---|---|---|
| Bioaccessibility | 98% (Pre-solubilized) | Variable (Requires Gastric Dissolution) |
| Assimilation Window | Immediate (1–4 Minutes) | Delayed (20–45 Minutes) |
| Mucosal Contact | Full Systemic Exposure | Limited (Isolated to lower GI release) |
| Excipient Load | Zero Binders or Fillers | High (Stearates, Glidants, Shells) |
| Molecular Stability | Cold-Processed Liquid | Risk of Heat Degradation (Compression) |
Maximizing Catechin Stability: To ensure the 72mg of EGCG reaches the small intestine with maximum integrity, we recommend:
- Empty Stomach Consumption: Reduces competition for intestinal transporters.
- Vitamin C Synergy: Research suggests Vitamin C stabilizes catechins in the non-acidic environment of the intestine.
- Temperature Sensitivity: Avoid boiling water, which can degrade sensitive polyphenol structures.
A Note on EGCG Pharmacokinetics
In studies of green tea catechins, it is well established that EGCG is a low-permeability molecule (1). Regardless of the delivery format, only a fraction of ingested catechins will achieve systemic bioavailability.
However, for systemic transport to occur, the molecule must first reach the small intestine in a solubilized state. Solid capsules and tablets often suffer from “Dissolution Lag,” where the EGCG remains trapped in the supplement matrix as it passes through the primary absorption window (2).
The ChiTea Advantage is not about overriding the laws of biology, but about optimizing the Net Payload. By delivering a pre-solubilized, bioaccessible dose, we remove the “Solubility Barrier,” ensuring that the highest possible percentage of our standardized 72mg EGCG is ready for uptake.
VIEW FULL LABORATORY REPORTS & COA
Independently verified for purity, potency, and 95% polyphenol standardization.
FAQs
Q: What is the difference between Bioavailability and Bioaccessibility? A: Bioaccessibility refers to the amount of a nutrient that is released from its food matrix (or capsule) and rendered soluble in the digestive tract, making it available for absorption. ChiTea’s liquid protocol ensures 98% bioaccessibility by delivering EGCG in a pre-solubilized state, bypassing the “dissolution barrier” of solid tablets.
Q: Why is 72mg of EGCG in liquid form considered highly effective? A: Clinical efficacy depends on the “Net Dose” that reaches the small intestine. Because liquid extracts avoid the binders and compression heat of capsules, the molecular integrity of the 72mg dose is preserved, and its immediate solubility maximizes the potential for intestinal transport.
Q: Does Vitamin C improve the stability of ChiTea? A: Yes. Research indicates that catechins like EGCG are sensitive to the non-acidic environment of the small intestine. Consuming ChiTea with Vitamin C or a slightly acidic carrier helps stabilize these polyphenols, further protecting the 72mg payload during the absorption window.
Scientific References & Clinical Citations
1. Intestinal Transport and Metabolism of Green Tea Catechins: A study on the permeability challenges of EGCG in the human GI tract. [PubMed 20138243]
2. Bioavailability of Green Tea Polyphenols: A comprehensive review of formulation influence on catechin uptake. [MDPI Nutrients 2020]
3. Effect of Formulation on Solubility: Research detailing how solubility directly impacts the bioavailability of EGCG payloads. [PubMed 22008148]
